EULAR 2025 Update for Behçet’s syndrome Save
EULAR has updated its recommendations for the management of Behçet’s syndrome, the last being published in 2018. The current update carries no recommendations from the previous version: seven recommendations have content modifications, four have minor wording amends, and one is entirely new.
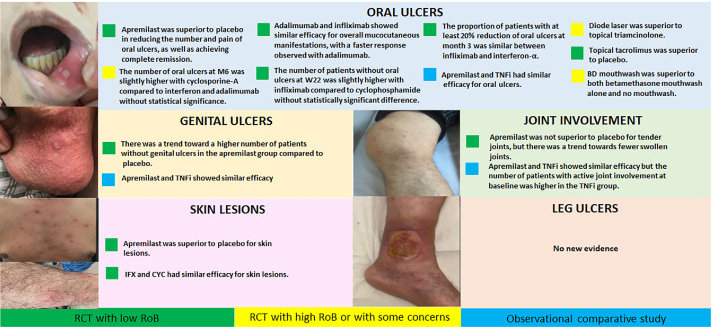
A systematic literature review included 7128 articles, 16 studies (7 randomised controlled trials [RCTs]) reported on mucocutaneous involvement, joint involvement, overall disease activity, or HRQoL.
The new recommendations includes five overarching principles and 12 recommendations. The overarching principles state that Behçet’s syndrome has a relapsing and remitting course that may be organ- or life-threatening, but that disease manifestations may ameliorate over time. As such, the goal of treatment is to prevent irreversible organ damage and to maximise quality of life. To support this, organ involvement should be evaluated throughout the disease course, and treatment should be individualised according to each person’s age, sex, type, and severity of organ involvement, disease duration, and their own personal preferences. Finally, Behçet’s syndrome requires a multidisciplinary approach and shared decision making, alongside patient education, adherence to treatment, and any lifestyle changes that are necessary to achieve optimal care.
The recommendations are tabulated according to the core manifestations of mucocutaneous, joint, eye, arterial, vascular, gastrointestinal, and parenchymal nervous system involvement. Each sets out the preferred first-line treatment approach, with some also specifying diagnostic measures. In brief, apremilast and immunosuppressives such as TNF inhibitors are recommended for refractory mucocutaneous and joint disease. For those with organ involvement, the recommendation is to achieve rapid remission with more aggressive treatment with glucocorticoids and immunosuppressives, and early use of monoclonal antibodies against TNF is encouraged where there are organ- or life-threatening manifestations.
The most important changes from the 2018 version are the preference for monoclonal TNFi in patients with eye, vascular, or nervous system involvement – both for induction of remission of the current attack, and as maintenance to prevent relapse.
EULAR hopes the updated recommendations will provide guidance for all stakeholders involved in managing Behçet’s syndrome, and ultimately help to improve the quality of care for people with this disease.
- BS has a relapsing and remitting course that may be organ- or life-threatening, while disease manifestations may ameliorate over time.
- The goal of treatment is to prevent irreversible organ damage and to maximise HRQoL.
- Organ involvement should be evaluated throughout the disease course, and mimickers should be ruled out with appropriate modalities.
- Treatment should be individualised according to age, sex, type, and severity of organ involvement, disease duration, and patient preferences.
- A multidisciplinary approach, patient education, shared decision making, adherence to treatment, and lifestyle changes are necessary for optimal care.
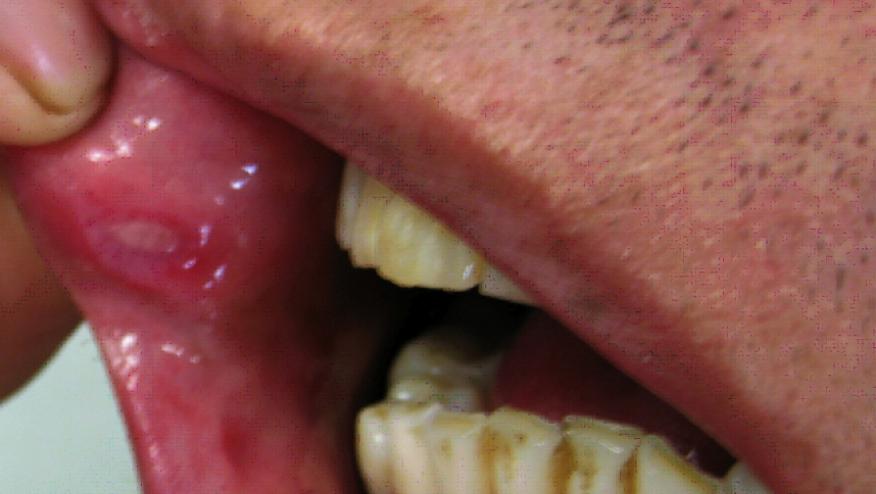
- Colchicine should be the first-line treatment for recurrent mucocutaneous lesions. In patients refractory or intolerant to colchicine, apremilast or TNFαi should be considered
- Topical measures such as glucocorticoids can be used for the management of oral and genital ulcers; chronic use of systemic glucocorticoids should be avoided
- Colchicine should be the first-line treatment for acute arthritis. Immunosuppressives should be considered in recurrent and chronic cases
- immunosuppressive treatment must be given in all patients with Behçet’s uveitis with the aim of inducing and maintaining clinical and angiographic remission (LoE 2). Monoclonal anti-TNFα antibodies, preferably infliximab in combination with other immunosuppressives, should be used in those with sight-threatening inflammation involving the posterior segment (LoE 5). Glucocorticoids should not be given as monotherapy
- For the management of pulmonary and peripheral artery aneurysms, high-dose glucocorticoids and infliximab are recommended; cyclophosphamide may be an alternative. Glucocorticoids should be slowly tapered and immunosuppressives, preferably monoclonal anti-TNFα antibodies, should be continued as maintenance.
- Vascular procedures, if necessary, should not be delayed following the prompt initiation of medical treatment. For patients with pulmonary artery aneurysms at high risk of major bleeding, embolisation should be preferred to open surgery
- For the management of acute thrombosis of deep veins, including cerebral venous sinuses, glucocorticoids and immunosuppressives, preferably monoclonal anti-TNFα antibodies should be considered. Immunosuppressives should be continued as maintenance.
- Anticoagulants may be added, provided the risk of bleeding is low and coexistent pulmonary artery aneurysms are ruled out.
- On cerebral venous sinus thrombosis with vision-threatening intracranial hypertension, surgical interventions should be given prompt consideration.
- Diagnosis, assessment of severity, and management of gastrointestinal involvement should be based on endoscopy.
- In patients with gastrointestinal involvement, 5-aminosalicylic acid (5-ASA) or azathioprine should be used with or without glucocorticoids. In severe or refractory patients, monoclonal anti-TNFα antibodies should be considered.
- For active parenchymal involvement, high-dose glucocorticoids and immunosuppressives, preferably infliximab, should be initiated. Glucocorticoids should be slowly tapered (LoE 5) and immunosuppressives continued as maintenance.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.