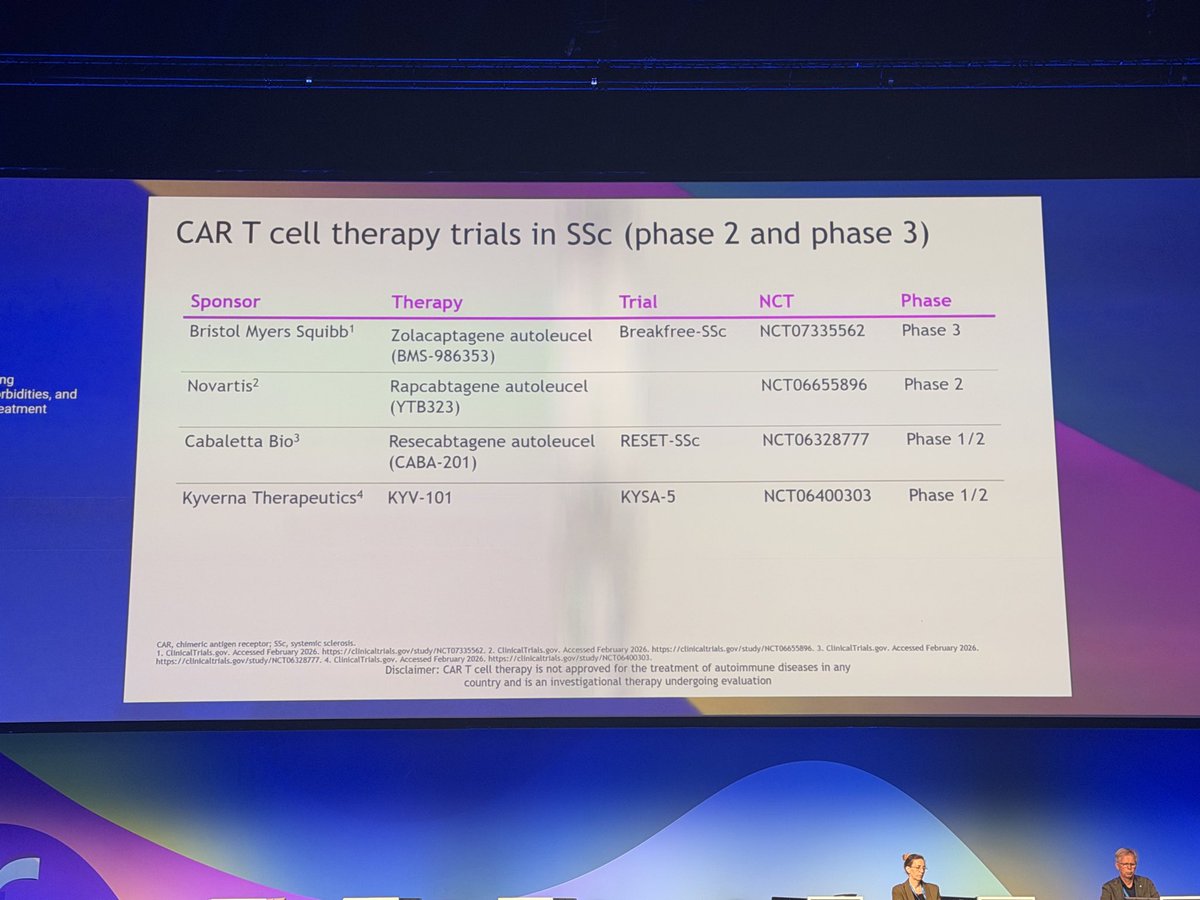
Rheumatology Shortage Cliff in Australia & USA
Australia is facing a deepening rheumatology manpower crisis with direct parallels to the situation in the United States.
Read Article

Dr. John Cush @RheumNow( View Tweet )

Dr. John Cush @RheumNow( View Tweet )

Dr. John Cush @RheumNow( View Tweet )


Jiha Lee @JihaRheum( View Tweet )

Dr. John Cush @RheumNow( View Tweet )
Links:


Dr. John Cush @RheumNow( View Tweet )
Australia is facing a deepening rheumatology manpower crisis with direct parallels to the situation in the United States.
Read Article

A cross-sectional study of US adults with systemic lupus erythematosus (SLE) shows significantly increased prevalence of polypharmacy, related to age, disease outcomes and often, non-SLE common medications.
A

Dr. John Cush @RheumNow( View Tweet )

A small, pilot, phase 2b trial has shown that the combination of leflunomide and hydroxychloroquine (LEF+HCQ) was effective and safe in treating patients with active Sjögren's disease.
Read ArticleXu et al. from Shanghai University have written an analysis of global lupus nephritis (LN) clinical trials and call for innovation.
Read Article

Mark Cuban @mcuban( View Tweet )

Dr. Jack Cush talks of birthdays, Quizzes and journal articles this week on the podcast.
Read ArticleUCB has released the Week 16 results from BE BOLD trial that will be presented at EULAR 2026 in London on Saturday, June 6. This head-to-head trial demonstrates that at week 16, bimekizumab (BKZ) yielded superior ACR50 results compared to risankizumab (RKB) adults with active psoriatic
Read Article
With their publication in June 2022 (1), the 3rd iteration of the Group for Assessment of Psoriasis and Psoriatic Arthritis (GRAPPA) Treatment recommendations for Psoriatic Arthritis (PsA) may have set a record or sorts.
By downloading this material, I acknowledge that it may be used only for personal use and personal education and that I will accredit RheumNow.com as the source and owner of this material. Commercial use or mass reproduction of this material without permission from RheumNow (info@rheumnow.com) is prohibited.