A Patient’s Plea for a New Paradigm in Autoimmune Disease
A current article in Nature Reviews Rheumatology has a patient boldly asking why we rheumatologists aren't more like her oncologists?
Read ArticleA current article in Nature Reviews Rheumatology has a patient boldly asking why we rheumatologists aren't more like her oncologists?
Read Article
Malignancy rivals cardiovascular disease as a leading cause of death in patients with systemic autoimmune diseases. Chronic inflammation and immune dysregulation can drive oncogenesis, while antitumor immune responses can trigger autoimmune phenomena (paraneoplastic syndromes, checkpoint
Read ArticleA retrospective target trial emulations has shown that low-dose aspirin (ASA) given with a giant cell arteritis (GCA) diagnosis is associated with a lower risk of major adverse cardiovascular events (MACE), but a higher risk of GI bleeding.
Read Article
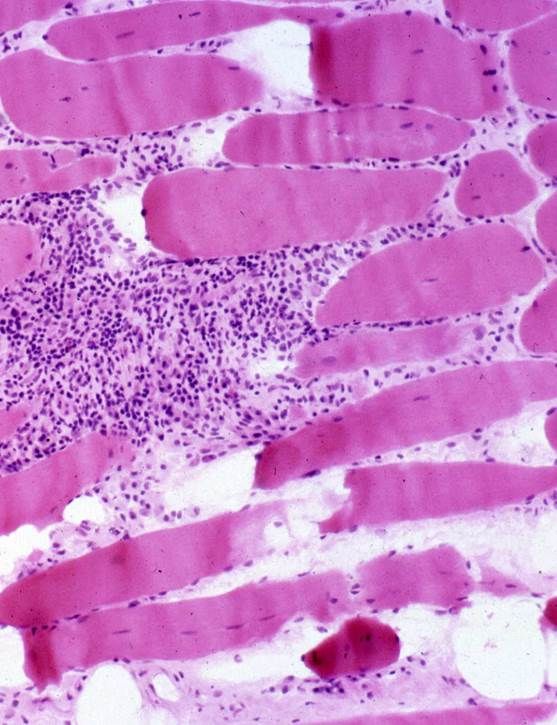
An Italian retrospective study of 411 idiopathic inflammatory myopathies (IIM) patients applied IMACS criteria to assess the risk of cancer and found significantly more cancers in those at high risk.
Read Article
Dr. Jack Cush reviews the news and journal reports from RheumNow.com this week - including his top four favorite subjects.
Read Article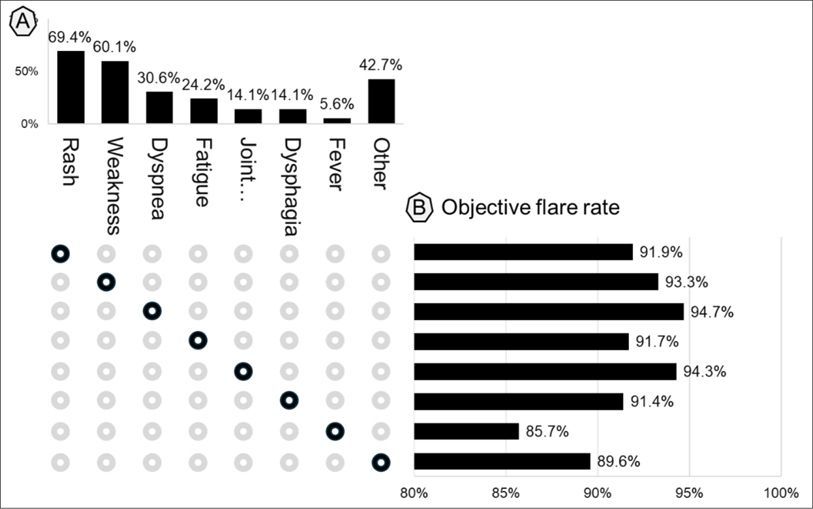
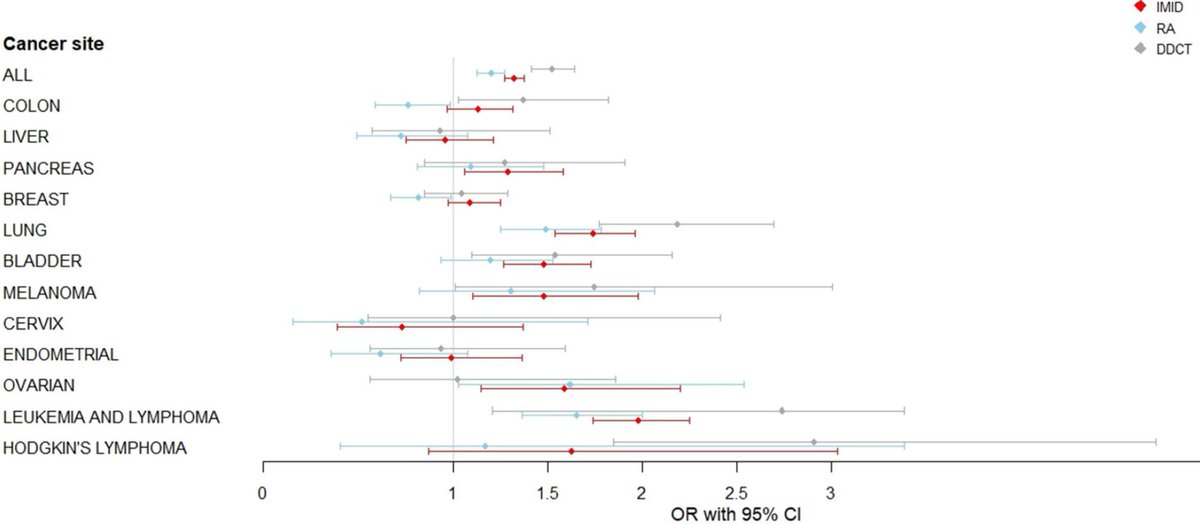
The risk of malignancy in rheumatoid arthritis (RA) patients receiving biologic agents was evaluated by metanalysis of randomized controlled trials (RCTs) and found no significant increased risk of malignancy compared with other disease-modifying antirheumatic drugs (DMARDs) or with placebo
Read Article


Dr. Jack Cush reviews the regulatory actions, news and journal reports from this past week on RheumNow.com.
Read Article

A retrospective study of patients who developed rheumatic immune-related adverse events (R-irAEs) after receiving immune checkpoint inhibitors (ICIs) at two oncology centers in Spain suggests that early recognition can lead to effective management that allows continuation of cancer therapy.
Read Article
Dr. Jack Cush reviews the news and journal reports from this past week on RheumNow.com.
Read Article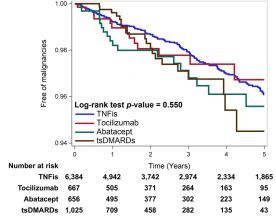
Dr. Jack Cush reviews the news and journal reports from this past week on RheumNow.com. Highlights include referral rules, combination biiologics in psoriasis and don't use JAK inhibitors in pregnancy.
Read Article
The quality of the meeting was on par with the host city, with extensive data presented on a range of topics, from social media to drug safety. The organization committee did a great job and I got the feeling that most people felt the congress was user friendly given the magnitude of the event. During this year’s meeting, I had the privilege of working with the RheumNow team, which gave me the opportunity to hone my social media skills and get my Twitter game on. After reviewing plenty of posters and going to numerous presentations, here are my top take home messages as classified by disease state.

By downloading this material, I acknowledge that it may be used only for personal use and personal education and that I will accredit RheumNow.com as the source and owner of this material. Commercial use or mass reproduction of this material without permission from RheumNow (info@rheumnow.com) is prohibited.