All News
Baricitinib Therapy in Covid-19 Pneumonia
The NEJM has published the results of the ACCT-2 Study, a double-blind, randomized, placebo-controlled trial evaluating baricitinib, an inhibitor of Janus kinase 1 (JAK1) and JAK2, plus remdesivir in hospitalized adults with Covid-19. The primary outcome was the time to recovery.
Read ArticlePaucity of Effectiveness Research on Telemedicine in RA
A systematic review of telehealth in delivering equivalent care in RA patients demonstrates there is limited evidence that virtual RA care is equivalent to conventional face-to-face care. Despite current widespread use, there is a great need for credible well-designed research that will address comparable patient outcomes, implementation strategies and long‐term health system consequences.
Read ArticleNEJM: Interleukin-6 Inhibition in Critically-Ill COVID-19
The pendulum and jury are wavering with regard to the benefits of interleukin-6 receptor antagonists in COVID-19; the current NEJM reports the results of a study favoring such biologic therapy in in critically ill COVID-19 patients.
Read ArticleMAXIMISE Trial - Secukinumab Efficacy in Psoriatic Spondylitis Patients
The results of the novel MAXIMISE trial have been published, demonstrating that secukinumab significantly improved axial signs and symptoms in psoriatic arthritis patients with axial manifestations with inadequate NSAID response.
Read ArticleRheumNow Podcast – Sweet Lies or Hard Truths? (2.26.2021)
Dr. Jack Cush reviews and discusses the news and Journal reports from the past week on RheumNow.com.
Read ArticleMeasureable Benefits to Treat-to-Target Management in RA
Desai and colleagues have published that when a learning collaborative group focused on a treat‐to‐target approach was compared to usual care, a modest but significant impact on implementation, treatment changes and patient outcomes in RA patients managed with a treat‐to‐target approach was seen.
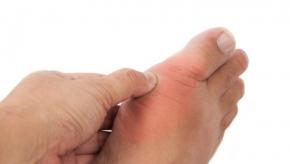
Read ArticleANAGO Study - Anakinra for Gout Flare
The interleukin (IL)-1 receptor antagonist anakinra (Kineret) was not superior to triamcinolone for relieving gout flares, but had similar effects for pain reduction as the glucocorticoid, a randomized phase II study found.
Read ArticleLung Preservation with Tocilizumab in Early Systemic Sclerosis
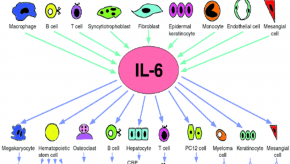
Arthritis & Rheumatology has published the results of a subanalysis from tocilizumab (TCZ) clinical trials showing that Il-6 inhibition was associated with preservation of lung function patients with early systemic sclerosis (SSc).
Read ArticleAvacopan - Is it Really a Win for ANCA-Associated Vasculitis?
The NEJM has reported the results of a study showing that avacopan, a C5a receptor inhibitor, is as effective as steroids at treating patients with antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis (AAV).
Read ArticleWeekly Semaglutide for Weight Loss in Adults
NEJM has reported the results of a trial showing semaglutide once weekly plus lifestyle intervention was associated with sustained, weight loss in overweight or obese adults; such that nearly one-third lost over 20% of their body weight.
Read ArticleACR Promotes New COVID-19 Vaccine Clinical Guidance for Rheumatic Patients
The American College of Rheumatology has published a draft guidance on the use of COVID-19 vaccination in rheumatic disease and musculoskeletal disease patients, based on the efforts of the North American Task force. The document provides guidance to rheumatology providers on the use of the COVID-19 vaccine and the associated management of RMD patients around the time of vaccination. Here is a summary of the recommendations.
Read ArticleSynovial Tissue Signature Guiding Targeted IL-6 Therapy in RA
RNA sequencing and classification of RA subsets has proven useful. RNA sequencing-based stratification of RA synovial tissue showed stronger associations with clinical responses compared with histopathological classification. Additionally, for patients with low or absent B-cell lineage expression signature in synovial tissue, tocilizumab is more effective than rituximab.
Read Article
Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:

Links:

Links:
Dr. John Cush RheumNow ( View Tweet)


Links:
Dr. John Cush RheumNow ( View Tweet)