All News
Baricitinib Derailed by FDA Review
Eli Lilly and Co disclosed today that upon further discussions with the US Food and Drug Administration (FDA) there would be a delay in further regulatory decisions regarding baricitinib, a JAK inhibitor, that is being developed for use in rheumatoid arthritis (RA).
Read ArticleBenlysta FDA Approved for Sub-Q Use in Lupus
The US Food and Drug Administration (FDA) has approved a new subcutaneous formulation of Benlysta (belimumab) for use in active, autoantibody‑positive Systemic Lupus Erythematosus (SLE) patients.
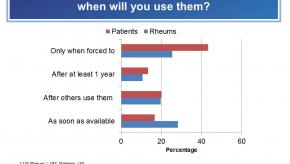
Read ArticleRheumatologists and Patients Concerns over Biosimilars - RheumNow “Live Vote” Results
The RheumNow “Live Vote” on the use, uptake and safety of biosimilars contrasts rheumatologist and patient views. Despite FDA approval and looming introduction, there is considerable concern, knowledge gaps and hesitancy regarding their uptake in the US. The survey's primary question demonstrates that less than one-third of US rheumatologists are prepared to adopt biosimilars when they are available.
Read ArticleThe RheumNow Week in Review – 21 July 2017
Dr. Jack Cush reviews the news from the past week on RheumNow.com:
Read ArticleObesity Impairs TNF Inhibitor Responses in Axial Spondyloarthritis
Investigators with the Swiss Clinical Quality Management (SCQM) program have studied the effects of obesity on patients with axial spondyloarthritis (axSpA) and, specifically, their response to tumor necrosis factor inhibitors (TNFi).
Read ArticleCurbside Consults - July 2017
Curbside Consults are cases submitted by our rheumatology colleagues as challenging therapeutic or safety issues. Answers are based on experience, with added evidence from the medical literature and published guidelines. Here are four new cases.
Read ArticleWithdrawal of TNF Inibitors Fails in Most RA Patients
The 12 month POET study analyzed 817 rheumatoid arthritis (RA) patients who achieved remission or low disease activity (LDAS) for ≥6 months and then randomized 2:1 to stopping or continuing their TNF inhibitor.
Read ArticleManaging Secondary Non-Response to Rituximab in Lupus
Rituximab, a chimeric anti-CD20 monoclonal antibody, has been effectively used in the treatment of multiple autoimmune disorders for over two decades. But the response to rituximab has been shown to not be as effective in SLE as it is in RA.
Read ArticleLinkage Between Gut and Joint Immunity in Rheumatoid Arthritis
Pianta, et al have published their findings in the Journal of Clinical Investigation showing that two novel autoantigens, GNS and FLNA, are highly expressed in synovium and share a sequence homology with gut microbesa; thereby providing a potential link between gut mucosal and joint immunity in rheumatoid arthritis.
Read ArticleBreastfeeding Rates Lower in Lupus
It is a common practice for rheumatologists to discuss medication-related teratogenicity and disease implications in case of pregnancy. Yet, discussion of breastfeeding are often omitted.
Read ArticleTildrakizumab, an IL-23 Inhibitor, is Successful in Plaque Psoriasis
Therapeutic options for cutaneous psoriasis (and psoriatic arthritis as well) are rapidly expanding. Lancet has published the impressive results of two phase III trials of tildrakizumab, a IgG1 antibody against interleukin 23 p19, in patients with active chronic plaque psoriasis.
Read ArticleACR/AAHKS Guidelines on Perioperative Management
The American College of Rheumatology has partnered with the American Association of Hip and Knee Surgeons to develop consensus-based guidelines for perioperative management of antirheumatic therapies for patients with rheumatic diseases who plan to undergo total joint replacement.
Read ArticleEULAR 2017 – Day 3 Highlights
Friday was a mega-day in Madrid as this day all the late-breaking abstracts were presented, in addition to the bulk of high-impact podium presentations in several areas.
Here are the major presentations from several clinical areas.
Read ArticleGuselkumab for Active Psoriatic Arthritis
A double-blind, placebo-controlled, multicenter study aimed to evaluate efficacy and safety of a fully human monoclonal anti IL-23 antibody guselkumab, in patients with active psoriatic arthritis.
Read ArticleThe RheumNow EULAR Week in Review – 16 June 2017
Dr. Jack Cush reviews some of the highlights from the past week at EULAR 2017 - June 16, 2017.
Follow RheumNow's expanded coverage from EULAR 2017 in Madrid.
Read ArticleLancet Launches EULAR 2017 and "A Platinum Age of Rheumatology"
The current Lancet edition for 10 June 2017 is a rheumatology rich collection.
Read ArticleSupreme Court Decision Favors Earlier Biosimilar Adoption
On Monday June 11th, the US Supreme Court unanimously ruled that biosimilar manufacturers can bring their drugs to market faster by eliminating the provision that the biosimar manufacturer had to give the innovator company 180 days notice before launching the new biosimilar.
Read ArticleFDA Approves Abatacept for Pediatric Use
Subcutaneous (SC) abatacept (Orencia) is now available for use in patients 2 years of age and older with moderately to severely active polyarticular Juvenile Idiopathic (JIA) according to Bristol-Myers Squibb.
Read ArticleSuspending Methotrexate for Influenza Vaccination
Vaccinating our rheumatoid arthritis patients against influenza every year is a safety priorty. However immunosuppressive therapies pose a challenge to vaccine administration as their use can hamper vaccine immunogenicity.
Read ArticleUpadacitinib Effective in Rheumatoid Arthritis
AbbVie released the preliminary results of a rheumatoid arthritis trial wherein its Jak inhibitor, upadacitinib, was tested against placebo, and shown to be superior in established RA patients who have failed prior DMARD therapy.
Read Article