All News
Does Evusheld shield people living with rheumatic diseases?
While COVID vaccination and subsequent booster remain the cornerstone, pre-exposure prophylaxis such as Evusheld (tixagevimab and cilgavimab) has been approved by FDA in December 2021 and EMA in March 2022 for people immunocompromised either due to a medical condition or receiving treatment with immunosuppressants and may not mount an adequate immune response to COVID vaccination. How does this therapy fare in RMD patients?
Read Article

Long COVID and Rheumatic Disease
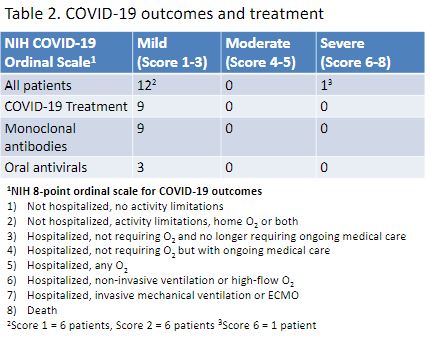
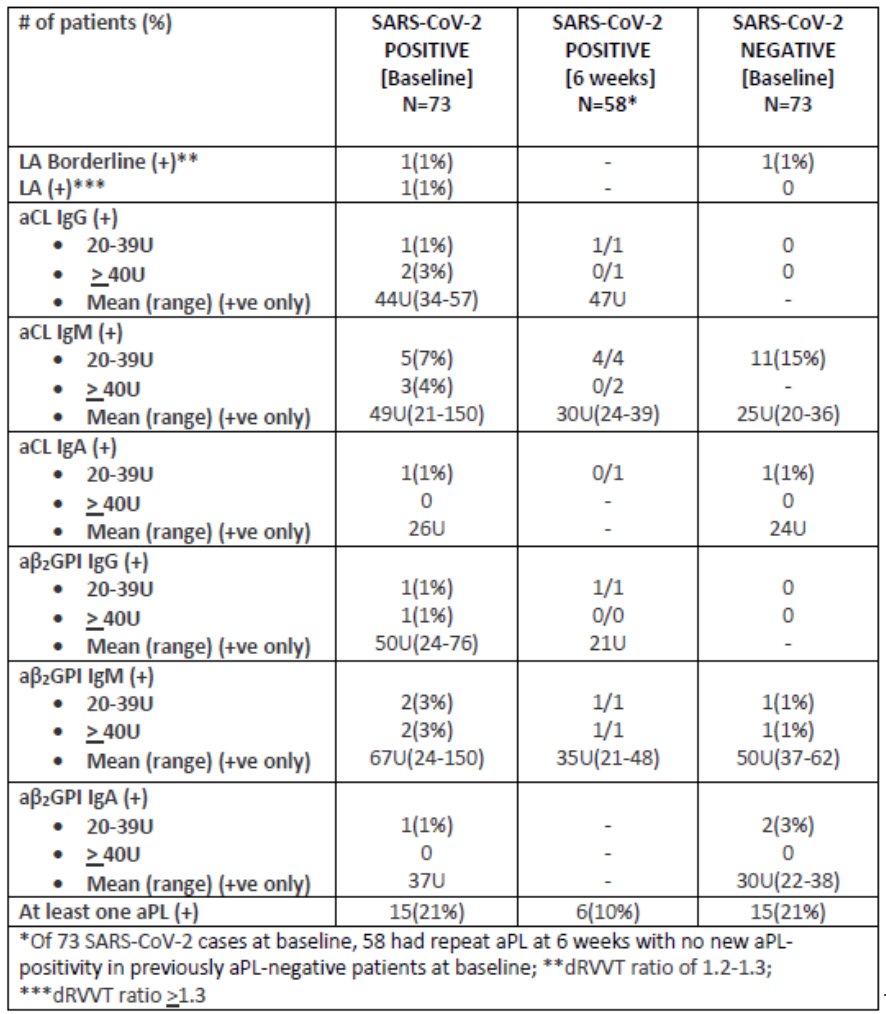
The COVID-19 pandemic has had a significant impact worldwide particularly among those who are immunocompromised. Numerous studies have highlighted the increased risk of acute SARS-CoV-2 infection among patients with rheumatic diseases (RD).
Read Article
Eric Dein ericdeinmd ( View Tweet)

Leonard Calabrese LCalabreseDO ( View Tweet)

TheDaoIndex KDAO2011 ( View Tweet)

Creaky Joints Presentations at ACR Convergence 2022
The patient advocacy organization CreakyJoints will present 13 scientific posters at this year's ACR Convergence 2022 and 8 posters as part of the patient perspectives track. Below is a sampling of these scientific posters.
Read Article
Catherine Sims, MD DrCassySims ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)
Links:
Links: