All News
Quiz Talk (5.22.2026)
Dr. Jack Cush talks of birthdays, Quizzes and journal articles this week on the podcast.
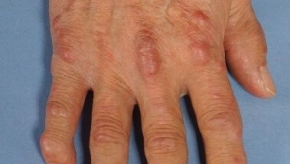
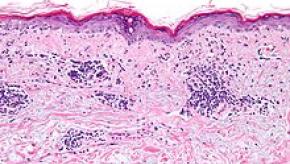
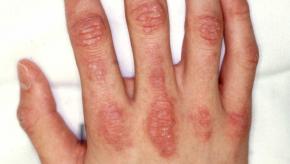
Read ArticleUpdate on Multicentric Reticulohistiocytosis
Have you ever seen multicentric reticulohistiocytosis? Don't worry, it's a rare non-Langerhans cell histiocytosis condition, characterized by papulonodular skin lesions and symmetric erosive polyarthritis, and has only been described in fewer than 500 cases reported worldwide.
Read ArticleToll-Like Receptor Drugs Effective in Cutaneous Lupus
Enpatoran, an investigational oral drug that inhibits Toll-like receptors (TLR) 7 and 8, was superior to placebo in relieving lupus skin manifestations in a dose-finding phase II trial.
Read ArticleInfluential Rheumatoid Factors (5.8.2026)
Dr. Jack Cush reviews the news and journal articles from this past week on RheumNow.com
Read ArticleBreaking the Rules: Dual-Advanced Combinations in Rheumatology
For decades, the standard of care in rheumatology has been combination therapy - pairing methotrexate (MTX) with an advanced biologic or synthetic agent to achieve optimal outcomes.
Read ArticleSwitch or Cycle - Upadacitinib vs Adalimumab in Refractory RA
After the first tumour necrosis factor inhibitor (TNFi) failure, patients with active rheumatoid arthritis (RA) responded by switching to upadacitinib, compared to cycling to a second TNFi, adalimumab.
Follow the Money (4.23.2026)
Dr. Jack Cush follows the money and all the news that fits the Rheumatology Gab for this past week.
Read ArticleStill's Disease Update
Arthritis Research & Therapy has published an overarching review of Still's disease - claiming it to be a single acquired and complex autoinflammatory disease in which both pediatric and adult forms share core pathogenic mechanisms, genetic associations and clinical presentations, with minor differences between children and adults who are afflicted.
Read ArticleTop Four and More (4.10.2026)
Dr. Jack Cush reviews the news and journal reports from RheumNow.com this week - including his top four favorite subjects.
Read ArticleMalignancies Not Increased with Biologic Therapies
The risk of malignancy in rheumatoid arthritis (RA) patients receiving biologic agents was evaluated by metanalysis of randomized controlled trials (RCTs) and found no significant increased risk of malignancy compared with other disease-modifying antirheumatic drugs (DMARDs) or with placebo.
Read ArticleJAMA Guideline on the Management of Crohn Disease
JAMA has published a current Clinical Guidelines Synopsis on the Management of Crohn Disease (CD) in Adults.
Read ArticleUpdated CRA/SPARCC Recommendations for Axial Spondyloarthritis
In 2024, the Canadian Rheumatology Association (CMA) and the Spondyloarthritis Research Consortium of Canada (SPARCC) published a comprehensive set of 56 treatment recommendations for the management of axial spondyloarthritis. This first update is focused on the reassessment of IL-17i recommendations.
Read ArticleWhat’s New PsA? (4.3.2026)
Dr. Jack Cush reviews the news and major articles from this week, including FDA safety warnings, reports from AAD, PsA trials.
Read ArticleBrepocitinib in Dermatomyositis
The NEJM reports that brepocitinib, an oral TYK2–JAK1 inhibitor, was studied in a phase 3 trial of refractory dermatomyositis (DM) patients and shown to have significant benefits at the higher dose.
Read Article
Dr. John Cush RheumNow ( View Tweet)



Dr. John Cush RheumNow ( View Tweet)