Predictors of Response in Lupus Save

A systematic review suggests that traditional measures (serologies and disease activity) are superior to biomarkers in predicting response to biologics in systemic lupus erythematosus (SLE) patients.
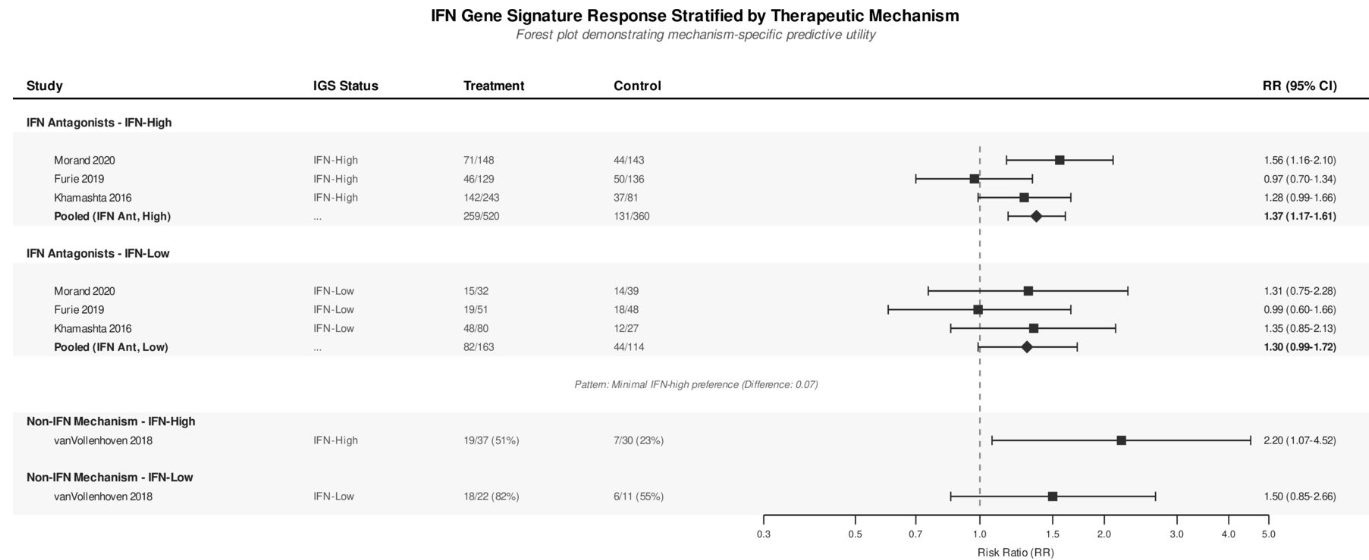
A systematic review identified 31 randomised controlled trials (RCTs) evaluating biologics (anti-BAFF, JAK inhibitors, anti-interferon, anti-IL-12/23, BTK inhibitors and T-cell modulators) in adult SLE patients. Primary outcomes included SLE Responder Index-4 response and time-to-flare. They compared the potential biomarker, interferon gene signature (IGS), to serologic activity (anti-double-stranded DNA positivity and/or low complement) and high disease activity (Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) ≥10).
The analysis included 17 374 participants. Biologics improved SLE Responder Index-4 response (RR 1.26, 95% CI 1.19 to 1.33, p <0.0001). Clinical response correlations were best for:
Serologically active disease (RR 1.45, 95% CI 1.28 to 1.63, p<0.0001,NNT=6.5).
High baseline SLEDAI ≥10 (RR 1.22, 95% CI 1.13 to 1.32)
IGS was not a ignificant predictor (RR 1.39, 95% CI 0.97 to 2.00, p=0.074).
Serologically active disease and high baseline disease activity significantly predict biologic therapy response with zero heterogeneity. By contrast IGS showed inconsistent predictive value.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.