All News
RheumNow Podcast – Falling or Falling in Love?
Dr. Jack Cush reviews the best journal articles from the past week on RheumNow.com.
Read ArticleARTIC REWIND - Don't Half DMARD Therapy
The ARTIC-REWIND study examined rheumatoid arthritis (RA) patients in remission on conventional synthetic disease-modifying antirheumatic drugs (csDMARDs) to see if they could "step-down" their csDMARDs theray; but the study showed that 25% those who went on half-dose experienced disease fl
Read ArticleDrug Monitoring Ineffective with Infliximab Treatment
The predictive value of therapeutic drug monitoring (TDM) while on biologics is debateable; gastroenterology routinely relies on TDM, while rheumatologists do not.
Read ArticleACR Updates COVID-19 Vaccine Guidance- April 2021
The ACR COVID-19 Vaccine Clinical Guidance was originally published on February 8, 2021 and updated on March 4, 2021. On April 28, 2021, new recommendations were added regarding mycophenolate, methotrexate, acetaminophen, and NSAID timing considerations,
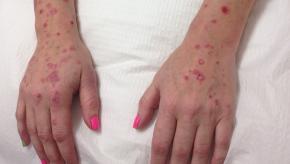
Read ArticleDual IL-17 Targeting is Effective but has More Candidiasis
Bimekizumab (BIM) is a monoclonal antibody that targets both interleukin-17A and interleukin-17F, which as a biologic target has worked well in the management of psoriatic disease. The current issue of the NEJM has two reports wherein BIM was shown to be more effective than secukinumab and adalimumab in treating patients with moderate-to-severe plaque psoriasis; however, safety concerns arise.
Read ArticleGolimumab Effective in non-radiographic Axial Spondyloarthritis
The GO-AHEAD study was designed to assess the long-term efficacy and safety of golimumab (GLM) in non-radiographic axial spondyloarthritis (nr-axSpA). After the 16-week, placebo [PBO]-controlled double-blind phase, 189/198 (95.5%) entered the long-term extension (OLE) for 36 weeks wherein G
Read ArticleBSRBR: Demyelinating Disease Following Anti-TNFα Therapy
Analysis of patients receiving anti–tumor necrosis factor α (TNFi) therapy in the British Society for Rheumatology Biologics Register (BSRBR) shows that the risk of demyelination is rare but is marginally increased by taking TNFi therapy.
Read ArticleDMARD Dropouts During COVID-19
A full year from the onset of the SARS-COV-2 pandemic, rheumatologists are still seeing the consequences of COVID, with many patients avoiding usual care, follow-up visits, routine lab testing and with many self-stopping their antirheumatic meds, because it seemed like the right thing to do.
Read ArticleRheumatology Care Derailed by COVID-19
A survey study from the ArthritisPower PPRN or CreakyJoints database shows that during the COVID-19 pandemic patient care was substantially affected in unforeseen ways.
Read ArticlePsoriatic Disease Management - RNL 2021 Highlights
This year’s coverage of psoriatic arthritis was once again, both exciting and informative. Topics included an overview of the latest therapeutic options, an excellent discussion on psoriasis specifically, and the epidemiology of psoriatic arthritis.
Read ArticleSafety of Paternal DMARD or Biologic Use
A large cohort study of expectant fathers treated for immune-mediated inflammatory diseases (IMIDs) failed to show any detrimental effect from paternal exposure to immunosuppressive or biologic agents on offspring outcomes.
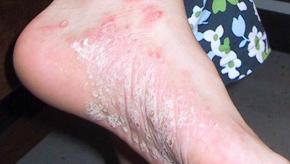
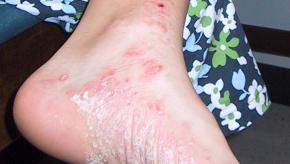
Read ArticleApremilast Equals MTX in Palmoplantar Psoriasis
Palmoplantar psoriasis (PPP) is a therapeutically challenging variant of psoriasis; nonetheless, a prospective, randomized, active-controlled trial from India suggests that apremilast is effective and safe in patients with PPP.
Read ArticleRheumNow Podcast – Pounds of Prevention (3.5.2021)
Dr. Jack Cush reviews and discusses nearly a dozen news reports and journal articles from the past week on RheumNow.com.
Read ArticleMeasureable Benefits to Treat-to-Target Management in RA
Desai and colleagues have published that when a learning collaborative group focused on a treat‐to‐target approach was compared to usual care, a modest but significant impact on implementation, treatment changes and patient outcomes in RA patients managed with a treat‐to‐target approach was seen.
Read Article
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)
Links:

Ruth Dobson drruthdobson ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)

Links:
Dr. John Cush RheumNow ( View Tweet)