Cost-Effective Use of Biological and Targeted Synthetic DMARDs Save
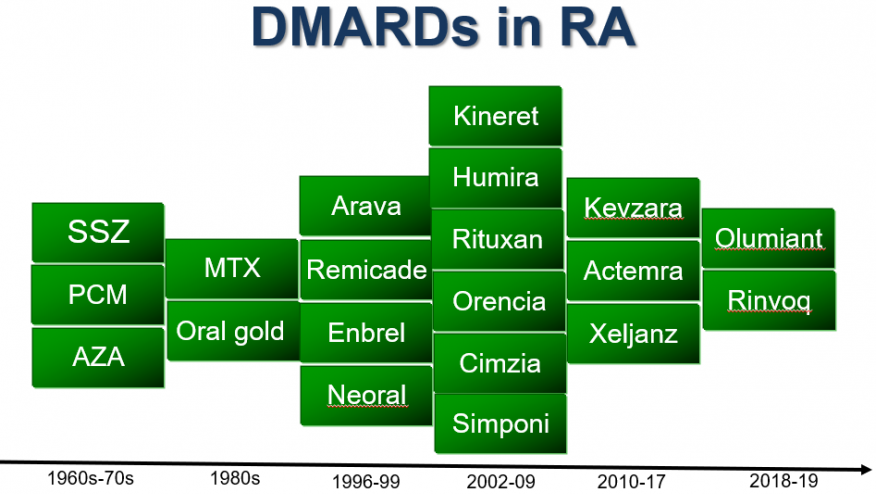
An international task force of 13 European experts have developed evidence-based points to consider regarding the cost-effective use of biological and targeted synthetic disease-modifying antirheumatic drugs (b/tsDMARDs) in the treatment of inflammatory rheumatic diseases (including rheumatoid arthritis, psoriatic arthritis and axial spondyloarthritis).
Twelve identified strategies for cost-effective use were identified and followed with a medical literature review that resulted in 30 systematic reviews and 21 RCTs upon which a set of overarching principles and points to consider were formulated by Delphi procedure.
Overall, the authors present 5 overarching principles and 20 final points to consider, 12 of which addressed cost-effective use of b/tsDMARDs.
Overarching principles
- Treatment choices must be based on shared decision-making between the patient and the rheumatologist.
- Treat-to-target is the cornerstone of b/tsDMARD-based treatment in RA, PsA and axSpA.
- Cost-effectiveness considerations are an important aspect of treatment, and rheumatologists should have a leading role regarding this.
- Reimbursement policies should cover cost-effective use of pharmacological treatments, both on-label and off-label, when it is evidence based and supported by (inter)national guidelines.
- Bio-originators and biosimilars are considered similar, and thus all recommendations apply equally to bio-originators and biosimilars.
Points to consider
Response prediction
- Therapeutic drug monitoring of b/tsDMARDs in patients with RA, PsA and axSpA is not advised because of the absence of evidence on efficacy and safety.
- Using other predictors for either choosing or tapering a particular b/tsDMARD is not advised because none have demonstrated superiority to standard care.
- Drug formulary policy: Rheumatologists might consider to adopt and use a drug formulary for their practice, primarily based on effectiveness and safety, and cost-effectiveness thereafter.
- Biosimilar/generic drug use: A biosimilar, if approved by a drug-regulating authority in a highly regulated area, should be preferred if it is the most cost-effective version of the drug.
- A single transition from a bio-originator to one of its biosimilars should be considered if it contributes to the cost-effectiveness of the treatment.
- Avoid dose loading: When initiating abatacept or certolizumab in RA, or secukinumab in PsA or axSpA, rheumatologists might consider to initiate treatment using the maintenance dose, as dose loading has not shown superior efficacy
- For the other b/tsDMARDs, there is no information on the effect of dose loading. Therefore, these drugs should be used as authorised.
- Initial lower dose: In RA, low-dose rituximab (1*1000 mg or 2*500 mg per cycle) has similar efficacy and less toxicity compared with authorised-dose rituximab (2*1000 mg) and should thus be preferred over the authorised dose.
- In patients with RA, rheumatologists might start with the lower dose§ of baricitinib or tocilizumab because of a more favorable safety and/or cost-effectiveness profile.
- Combination therapy: In patients with RA, rheumatologists should combine the b/tsDMARD with methotrexate to maximize efficacy; in patients who cannot use methotrexate as co-medication, IL-6 pathway inhibitors and JAK-inhibitors might be preferred over other bDMARDs.
- For patients with PsA or axSpA, combination therapy of a TNF inhibitor with methotrexate cannot be advised, because increased efficacy compared with TNF inhibitor monotherapy is not shown.
- For patients with PsA or axSpA, combination therapy of non-TNF inhibitors with methotrexate cannot be advised because of the absence of evidence on efficacy and safety.
- Route of administration: For patients with RA, non-inferiority of subcutaneous versus intravenous treatment of abatacept, infliximab and tocilizumab has been shown, and thus rheumatologists can choose the most cost-effective route of administration when initiating one of those drugs.
- For patients with RA, a single switch from subcutaneous to intravenous tocilizumab or vice versa did not affect efficacy or safety, and thus rheumatologists might consider this for cost-effectiveness reasons.
- Medication adherence: Rheumatologists should take adherence into account in the management of their patients by using the current points to consider** to manage non-adherence of b/tsDMARDs.
- Disease activity–guided dose optimization: For patients with RA in whom the treatment target is reached and sustained, rheumatologists should consider disease activity–guided dose optimisation of anti-TNF drugs.
- For patients with RA in whom the treatment target is reached and sustained, rheumatologists might consider disease activity–guided dose optimisation of IL-6 inhibitors, rituximab, baricitinib or abatacept.
- For patients with axSpA and PsA in whom the treatment target is reached and sustained, rheumatologists might consider disease activity–guided dose optimization of anti-TNF drugs.
- Rheumatologists can use any disease activity–guided dose optimization scheme, as none is preferential based on the evidence.
- Non-medical drug switching: Non-medical switching within or between b/tsDMARD classes is not advised because of the absence of evidence on efficacy and safety.









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.