Ixekizumab With Tirzepatide Efficacy in Obese Psoriatic Arthritis Save

The TOGETHER-PsA trial has demonstrated the efficacy and safety of ixekizumab (IXE) administered with tirzepatide (TZP) in adult active psoriatic arthritis (PsA) patients who were overweight or obese.
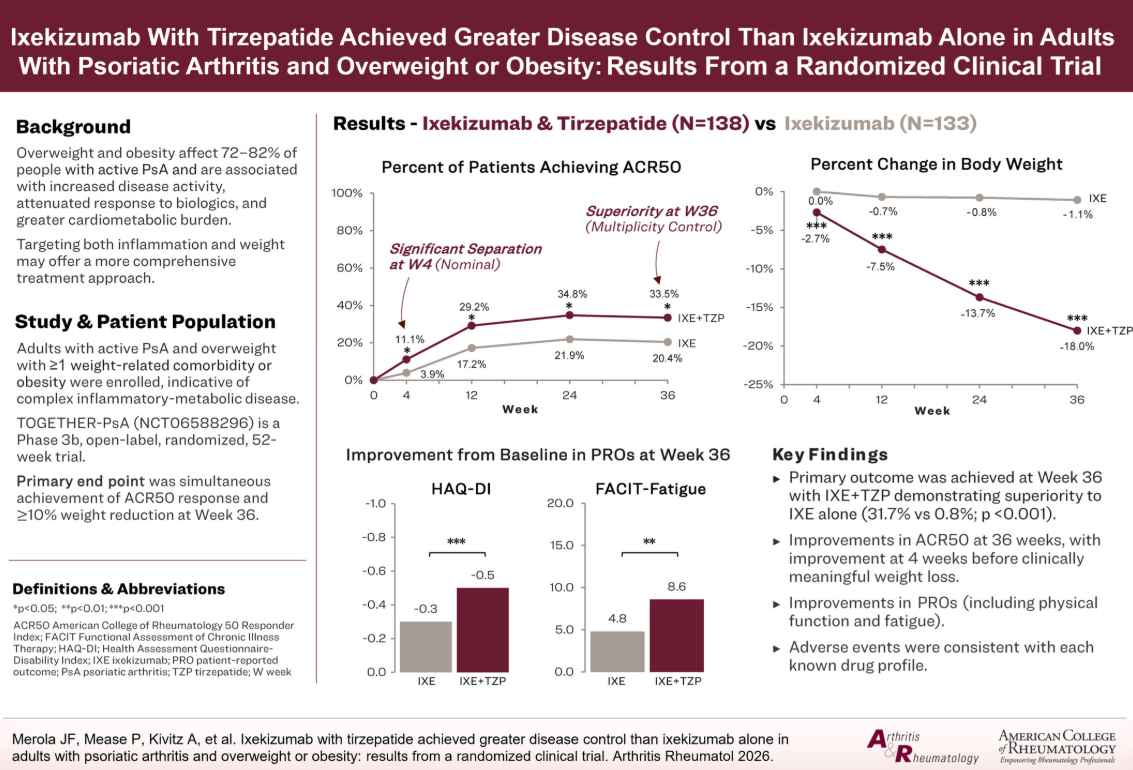
This trial (ClinicalTrials.gov NCT06588296) was a phase 3b, 52-week study in active PsA patients who were overweight (BMI ≥27 to <30) with at least one weight-related comorbidity or obesity (BMI ≥30). Patients were randomized to receive either IXE plush TZP or IXE alone. The primary end point was achieving and ACR 50 plus ≥10% weight reduction at 36 weeks.
A total of 271 participants were randomized. The primary end point favored the combination:
- IXE + TZP arm = 31.7%
- IXE alone = 0.8% (P < 0.001)
Other secondary endpoints favored IXE + TZP
- ACR50 responses (33.5% vs 20.% in IXE alone (P = 0.02).
- ACR20 (P < 0.001)
- MDA (minimal disease activity) (P < 0.05)
- Psoriasis Area and Severity Index score (P < 0.01)
- Health Assessment Questionnaire–Disability Index (∆ −0.2; nominal P < 0.001)
- FACIT (Functional Assessment of Chronic Illness Therapy–Fatigue) (improvement of 3.8; nominal P < 0.01)
No new safety signals were observed.
The combination of an incretin and IL-17 inhibitor lead to clinically meaningful improvement of PsA, physical function, weight reduction, and quality of life.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.