All News

The RheumNow Podcast is up – Should you Believe in Vitamin D or Rituximab? Watch it on RheumNow>> https://t.co/bX5u9KcKYz or listen to the podcast on iTunes or SoundCloud>> https://t.co/MxDcIyej3L
Dr. John Cush RheumNow ( View Tweet)

The RheumNow Podcast is up – Should you Believe in Vitamin D or Rituximab? Watch it on RheumNow>> https://t.co/bX5u9KcKYz or listen to the podcast on iTunes or SoundCloud>> https://t.co/MxDcIyej3L
Dr. John Cush RheumNow ( View Tweet)
RheumNow Podcast – Believe in Vitamin D or Rituximab? (9.20.19)
Dr. Jack Cush reviews the news and journal articles from the past week on RheumNow.com.
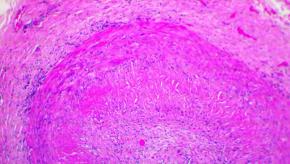
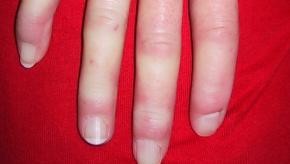
Read ArticleSensitivity of Temporal Artery Biopsy
Metanalysis shows that temporal artery biopsy (TAB) for the diagnosis of giant cell arteritis (GCA) has a sensitivity of 77%, similar to results seen with temporal artery imaging. These data suggest clinicians may be willing to accept a GCA diagnosis without proof by TAB.
Read ArticleFDA Grants Breakthrough Status for Potential Lupus Nephritis Drug
Obinutuzumab (Gazyva) has been granted Breakthrough Therapy Designation (BTD) by the U.S. Food and Drug Administration (FDA) for use in adults with lupus nephritis (LN). The drug made by Genentech, is going forward based on the Phase II NOBILITY study in adult patients with proliferative lupus nephritis (LN). Currently, there are no FDA-approved medicines for lupus nephritis.
Read ArticleRheumNow Podcast – The End of Arthritis (9.13.19)
Dr. Jack Cush reviews the news and journal articles from the past week on RheumNow.com.
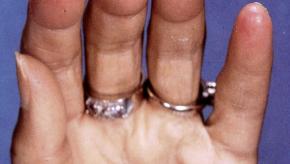
Read ArticleRiociguat Fails in Systemic Sclerosis-Associated Digital Ulcers
Riociguat is an oral, selective soluble guanylate cyclase stimulator that has been studied in patients with digital ulcers (DU) due to systemic sclerosis (SSc) but study results show that short term (16 weeks) riociquat therapy does not sufficiently reduce the DU burden in SSc patients.
Read ArticleNintedanib FDA Approved for Scleroderma Lung Disease
Last Friday, the US Food and Drug Administration approved Ofev (nintedanib) to slow the rate of decline in pulmonary function in adults with interstitial lung disease associated with systemic sclerosis or scleroderma, called SSc-ILD.
ILD as a complication of SSc may lead to progressive loss of lung function and may be associated with a significant mortality risk. Prior to the approval of Olev, there were no FDA approved drugs for SSc-ILD.
RheumNow Podcast – Antibiotics Increase RA Risk (8.16.19)
Dr. Jack Cush reports the news and important journal articles from the past week on RheumNow.com.
Read ArticleWith Autoimmunity, Checkpoint Inhibitors Can Be Used
Among patients with pre-existing autoimmune diseases who developed cancer and were treated with immune checkpoint inhibitors (ICI), flares of the underlying disease and other immune-related adverse events were common, a retrospective study conducted in France showed.
Read ArticleRheumNow Podcast – Tricked Up Lupus Criteria (8.9.19)
Dr. Jack Cush reviews the news and journal reports from the past week on RheumNow.com, including new SLE criteria, ULT in gout and MSU reductions, IVIG in ANCA vasculitis and non-TNF biologics outperform the TNF inhibitors.
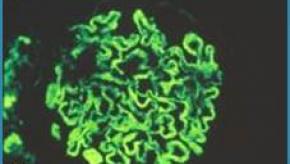
Read ArticleNew EULAR/ACR Classification Criteria for SLE
The European League Against Rheumatism (EULAR) and the American College of Rheumatology (ACR) have jointly developed new classification criteria for systemic lupus erythematosus (SLE); prompted by the need for criteria that were both highly sensitive and specific. The net result is improved sensitivity and specificity, but the use of positive ANA requirement along with a longer list of weighted criteria ensures its utility in SLE research (including early or latent SLE), but not clinical practice.
Read ArticleSjogren's Syndrome at Risk for Psychiatric Disorders
A population-based claims study from Taiwan shows significantly increased incidences of depressive disorder, anxiety disorder, and sleep disorder in patients with primary Sjögren’s syndrome (pSS).
Read ArticleAbatacept Disappoints in Systemic Sclerosis
A 12‐month, Phase 2 trial has shown that subcutaneous abatacept was well tolerated in patients with diffuse cutaneous systemic sclerosis (dcSSc), but failed to significantly change the skin outcomes as measured by the change in modified Rodnan skin score (mRSS)
Read ArticleRheumNow Podcast – FDA Hyperactivity (7.26.19)
Dr. Jack Cush reviews the news, journal reports, and FDA releases and deliberations covered this past week on RheumNow.com
Read ArticleFDA AAC Splits Vote in Favor of Nintedanib for Scleroderma Interstitial Lung Disease
The FDA convened Arthritis Advisory Committee to consider nintedanib for the treatment of systemic sclerosis-associated interstitial lung disease (SSc-ILD) and voted 10-7 in favor of approving the drug for this new indication.
Read ArticleFDA Approves Apremilast for Behcet's Oral Ulcers
The US Food and Drug Administration (FDA) has approved apremilast (Otezla) for the treatment of oral ulcers associated with Behçet’s Disease, a rare chronic multisystem inflammatory disease that affects about 5 in 100,000 persons in the USA.
Read ArticleCardiovascular Disease Increased in Hospitalized Lupus Patients
Systemic lupus erythematosus (SLE) patients who are hospitalized have an increased prevalence of atherosclerotic cardiovascular disease (ASCVD) and its individual phenotypes of coronary artery disease (CAD), peripheral artery disease (PAD), and cerebrovascular disease.
Read ArticleRheumNow Podcast – Boiling Hot MAS (7.12.19)
Dr. Jack Cush reviews this week's news and journal reports featured on RheumNow.com. PsA v. non-PsA pregnancies, Still's disease, lupus, nutritional supplements and dietary interventions, use of cannabis, tanezumab in OA, CBD oils and more.
Read ArticlePregnancy Outcomes Improve in Lupus
Pregnancy for patients with lupus has long been considered high risk and associated with both medical and obstetric complications, but outcomes have improved over the last 2 decades and continue to improve. The large decline in in-hospital maternal mortality was greater for lupus pregnancies than for non-lupus pregnancies. Findings from a retrospective cohort study are published in Annals of Internal Medicine.