Adjunctive Prednisolone for Kawasaki Disease Save
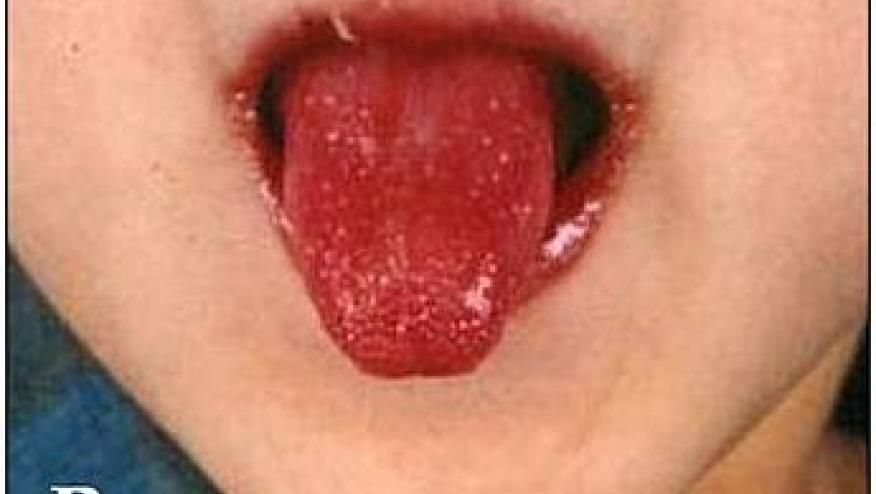
An NEJM study has shown that glucocorticoids provide no added benefit when added to standard primary treatment in Kawasaki disease patients.
An open-label, randomized, controlled trial in China enrolled and randomized 3208 newly diagnosed Kawasaki children receive prednisolone plus standard treatment or standard treatment alone. The primary outcome was the occurrence of coronary-artery lesions at 1 month after illness onset.
Baseline coronary-artery lesions were seen in 870 of 3184 (27.3%) patients. At 1 month, coronary-artery lesions developed in 16% on prednisolone vs 13.8% not on prednisolone (adjusted risk difference, 1.1 percentage points; 95% confidence interval, −1.0 to 3.4; P=0.31). At 3 months, the incidence of coronary-artery lesions was 12.6% with prednisolone plus standard therapy and 10.5% with standard treatment alone; the percentage of participants with progression of coronary-artery lesions was 28.6% and 28.9%, respectively.
Nonsignificant differences in secondary outcomes included:
- Rescue therapy (4.6% vs 10.1%)
- Duration of fever (8.4 hours vs 13.2 hours)
- C-reactive protein reductions at 72 hours (67.5 mg/L vs 59.8 mg/L)
- Decreases in coronary-artery z scores
- Medium-to-giant coronary-artery aneurysms (1.9% vs 1.1%).
- Adverse events
Addition use of prednisolone in the treatment for Kawasaki disease does not appear to be justified.
Continue Reading
ADD THE FIRST COMMENT
Disclosures
The author has no conflicts of interest to disclose related to this subject









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.