Romilkimab Effective in Early Diffuse Systemic Sclerosis Save
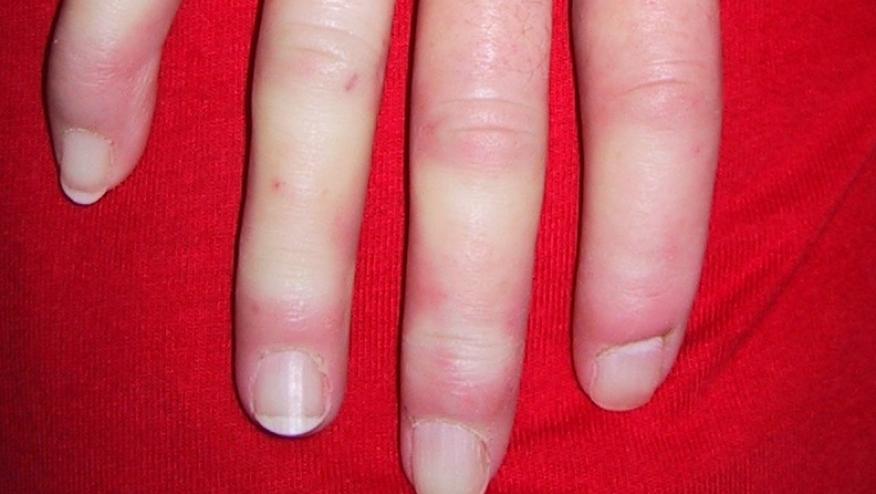
A phase II study of patients with early diffuse systemic sclerosis (SSc) has demonstrated improvement in SSc skin outcomes by targeting T cells and fibrosis with the bispecific monoclonal antibody romilkimab (RMK).
Romilkimab is an engineered, humanised, bispecific immunoglobulin-G4 antibody that binds and neutralises IL-4/IL-13 making it ideal for exploration in fibrosis.
This double-blind, proof-of-concept, phase II study enrolled total of 97 SSc patients and treated them for 24 weeks with either placebo or subcutaneous romilkimab 200 mg weekly. The primary endpoint was change in modified Rodnan skin score (mRSS) at week 24.
Change in mRSS favored RMK (–4.76 vs. –2.45) over placebo (p=0.0291, one-sided).
Adverse events were equal between groups and there were two deaths (one scleroderma renal crisis (romilkimab) and one cardiomyopathy (placebo)).
Serious adverse events were seen in the placebo group, where 2 patients had a cardiovascular events (one cardiac failure, one cardiomyopathy).
These early data suggest that RMK is capable of yielding significant skin benefits in early dcSSc and that further study is warranted.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.