Anakinra May Attenuate Stroke Outcomes Save
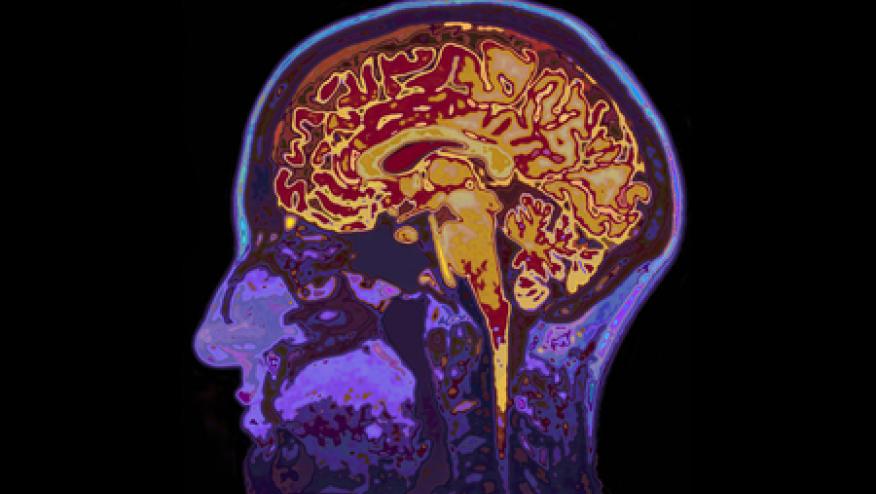
The journal Stroke has published a trial wherein the use of subcutaneous IL-1Ra (anakinra) was shown to reduce the peripheral inflammatory response in those with an acute ischemic stroke. (Citation source: http://bit.ly/2uoNXEL)
Based on data showing worse stroke outcomes in the face of high IL-6 and C-reactive protein (CRP) levels, researchers sought to assess whether control of inflammation post-CVA would limit cerebral ischemia and improve outcomes after ischemic stroke.
A double-blind, randomized, placebo-controlled phase 2 trial of subcutaneous anakinra (100 mg given twice daily for 3 days) was initiated within 5 hours of ischemic stroke onset. The primary outcome was difference in concentration of log(interleukin-6) as area under the curve to day 3. Secondary outcomes included the 3-month stroke outcome assessed by modified Rankin Scale.
A total of 80 patients (mean age, 72 years) were enrolled and 73% received intravenous thrombolytic therapy. Patients were then treated with either placebo or anakinra bid for 3 days. After 3 days the anakinra patients had significantly lower plasma interleukin-6 (P<0.001) and plasma C-reactive protein (P<0.001) levels.
Despite being well tolerated with no safety concerns, anakinra lowered the modified Rankin Scale, but this was not significant (OR=0.67; (0.29–1.52), P=0.34).
These insufficient 3 mos clinical outcomes may relate to the relatively short duration of anakinra therapy and other factors. Yet these data suggest the potential interplay between inflammation (IL-1) and tissue damage from thrombotic events.
These data follow a recent Lancet report of the CANTOS study showing cannakinumab (IL-1 inhibition) significantly lower rate of recurrent cardiovascular events (in high-risk CV patients); and at the same time reduce the rate of total cancer deaths, especially lung cancer.









If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.