A Pilot Trial of Anifrolumab in Discoid Lupus Save
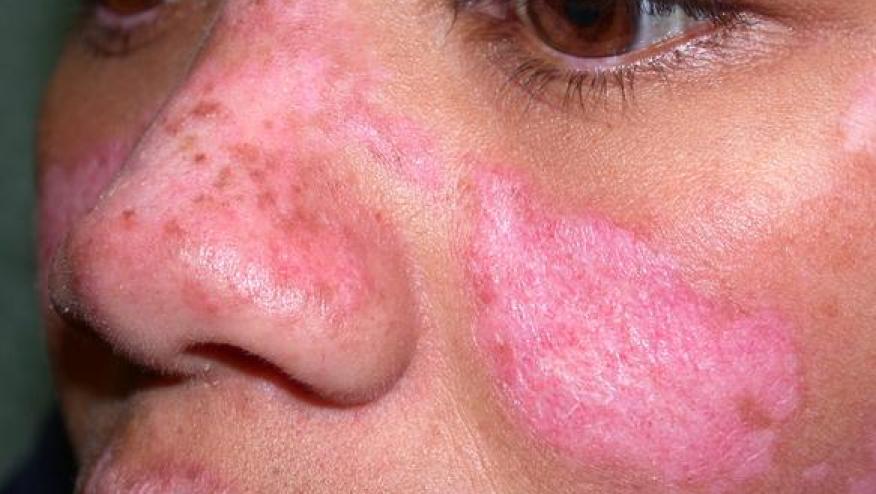
A research report in JAMA Dermatology describes the use of the type 1 interferon receptor inhibitor anifrolumab in patients with chronic discoid lupus erythematosus (DLE).
Anifrolumab is FDA approved since 2021 for use in adults with moderate-to-severe systemic lupus erythematosus (SLE), wherein it demonstrated significant benefits in lupus-associated skin disease.
Ten refractory DLE patients from the Brigham and Women’s Hospital and Massachusetts General Hospital were treated with anifrolumab, with 8 patients receiving at least 8 weeks of therapy (median age, 42.5 yrs). Clinical responses were assessed using the CLASI score (including both CLASI activity [CLASI-A], score 0-70 and CLASI damage [CLASI-D], score 0-56).
All patients showed significant improvement within 2 months of initiating anifrolumab. CLASI-A (activity) scores improved a mean of 65.1%, while CLASI-D (damage) scores improved 2.9%. Prior studies have suggested that a clinically meaningful improvement in DLE would be a 20% decrease in CLASI-A score.
This open label exploratory trial was notable for brisk and clinically meaningful responses, that would hopefully reduce the risk of permanent scarring. These early results are encouraging but require further well designed, controlled trials.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.