Vamorolone: a New Steroid on the Horizon? Save

Vamorolone is a potentially new alternative to traditional glucocorticoids for use in inflammatory diseases. It is a dissociated steroidal compound with reduced side effects, and in an animal model of inflammation has shown effective antiinflammatory properties and reduced systemic effects, as compared with prednisolone. But will this hold true in humans and at higher doses?
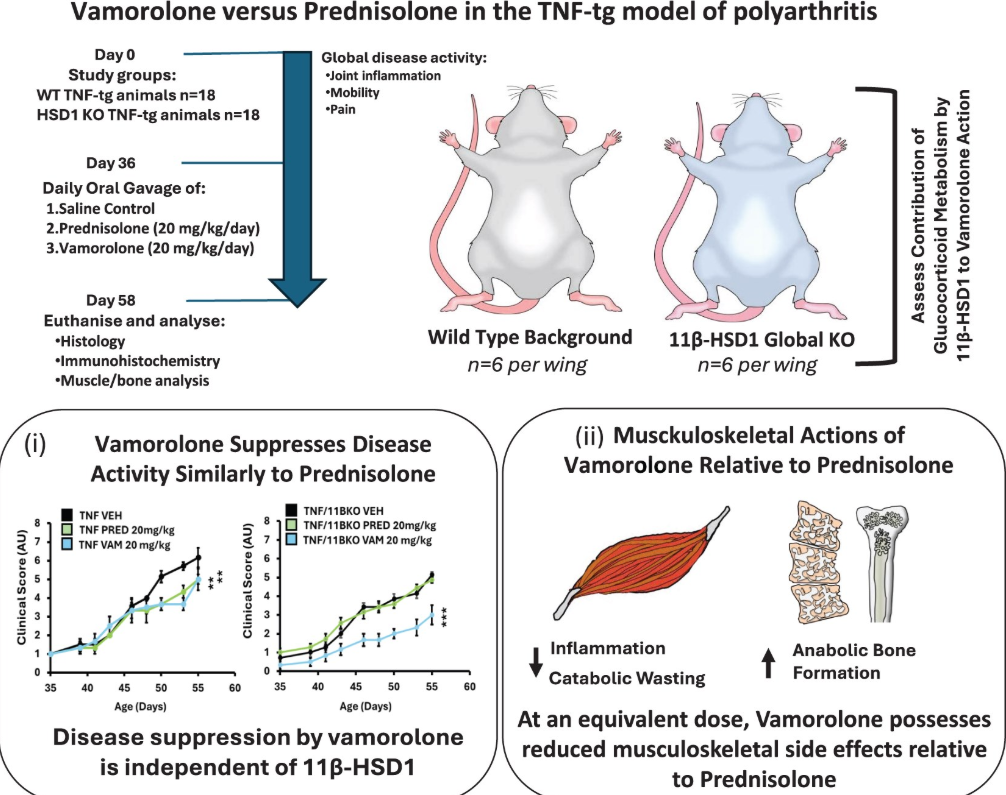
Vamorolone lacks the hydroxyl or ketone groups required for metabolism by 11β-hydroxysteroid dehydrogenase type 1 (11β-HSD1), a key enzyme that modulates glucocorticoid activity. This study examined vamorolone 11β-HSD1 resistance and assess therapeutic efficacy in the murine tumour necros factor-alpha-overexpressing (TNFtg) model of polyarthritis.
Vamorolone 20 mg/kg/day, prednisolone (standard of care) or vehicle were administered by gavage to TNFtg or TNFtg 11β-HSD1 knock-out (TNFtg11BKOKO) animals. Body weight and disease severity were scored daily, and markers of inflammation, joint destruction and side effects assessed at day 56 of age.
Vamorolone was entirely resistant to 11β-HSD1 metabolism in vitro. Vamorolone demonstrated comparable anti-inflammatory actions in TNFtg mice, with a comparable reduction in joint inflammation, serum interleukin-6 (IL-6) and synovitis, relative to prednisolone.
However, vamorolone-treated mice did not experience glucocorticoid side effects (adrenal atrophy, body weight reduction, muscle wasting or inhibition of anabolic bone metabolism). These benefits indicate that the efficacy of vamorolone is largely independent of 11β-HSD1 metabolism.
The FDA approved vamorolone in 2023 for use in children and adults with Duchenne muscular dystrophy (DMD). The drug is marketed under the name AGAMREE, and comes with all the usual warnings of GCs. The product label says most common adverse reactions (>10% ) are cushingoid features, psychiatric disorders, vomiting, weight increased, and vitamin D deficiency. While dosing depends on patient weight, the annual cost is approximately $79,000 USD (£62,812) per year. A systematic review and meta-analyis comparing vamorolone to standard GCs suggests the improved efficacy and safety of vamorolone for (but the external validity of these findings as well as the medication’s long-term effects remain to be determined).










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.