Proposed Cancer Screening for Inflammatory Myositis Patients Save
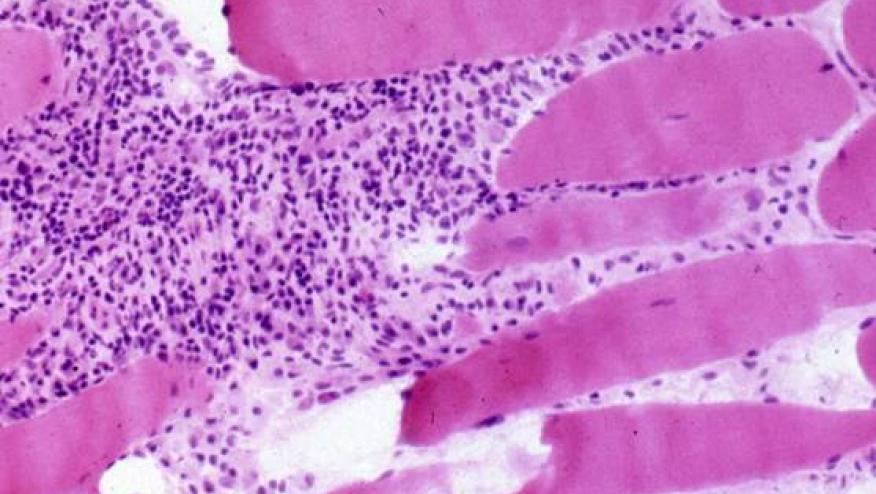
Today's plenary session features a novel proposal on how and which patients with Idiopathic Inflammatory Myopathy (IIM) should be screened for cancer. Abstract 0002 by Oldroyd et al, entitled "Cancer Screening Recommendations for Patients with Idiopathic Inflammatory Myopathy" took on this thorny practice issue.
While it is known that IIM patients may be at higher risk of developing certain cancers (especially lung, ovarian, colorectal, lymphoma, breast) during, before or after diagnosis, the certainty of this association isn't so high as to warrant cancer screening in all IIM patients.
The International Myositis Assessment and Clinical Studies Group (IMACS) studied this issue using an evidence- and consensus-based approach to develop a set of recommendations for IIM-associated cancer screening.
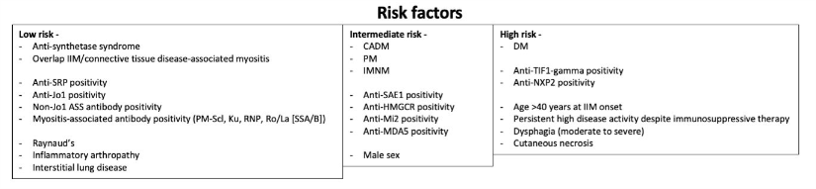
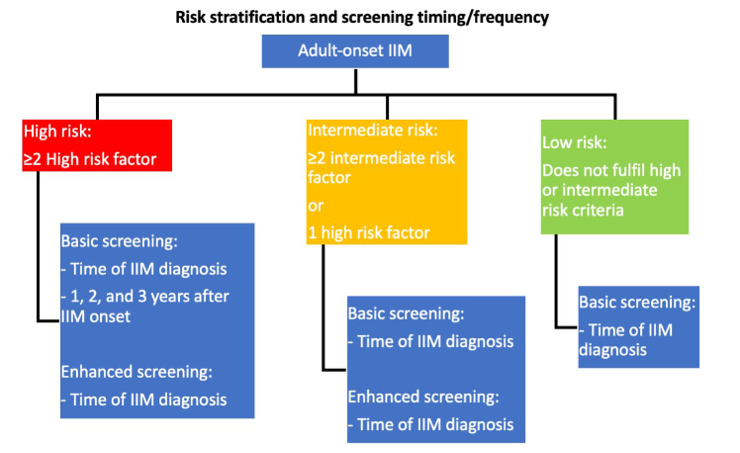
Their 18 recommendations (13 strong recommendations) are guided by stratifying IIM patients into low, intermediate, and high risk (for developing cancer) using myositis type, autoantibody status, and clinical features. Secondly, recommendations outline a “basic” screening panel (including chest X-ray radiography, basic laboratory blood tests) and an “enhanced” screening panel (including computed tomography, tumour markers). Thirdly, recommendations advise on the timing and frequency of screening via basic and enhanced panels, according to low/intermediate/high risk status (Figure 1). Recommendations also advise consideration of upper/lower gastro-intestinal endoscopy, naso-endoscopy, and 18F-FDG PET/CT scanning in specific populations.
Recommendations state that screening is not required for those with juvenile onset IIM (JDM) or inclusion body myositis. All should receive country, age and sex appropriate screening measures - basic screening (eg, history and exam, labs, ESR, CRP, UA, CXR, SPEP). Patients in intermediate and high risk groups should undergo enhanced screening and those with high risk should undergo enhanced screening every 3 years. Enhanced screening measures include gender specific assessments (CT scans, cervical screening, mammography, vaginal US, CA125, fecal occult blood) (all of these are "strong" recommendations).
Conditional recommendations include FDG/PET-CT for high risk patients in whom nothing is found or in high risk patients who are positive for TIF-1gamma and have
Recommendations
- They state that screening is not required for those with juvenile onset IIM (JDM) or inclusion body myositis.
- All newly diagnosed IIM patients undergo testing for myositis-specific autoantibodies (eg, Jo-1, PL-7, etc) and myositis asociate autoantibodies (eg, NXP-2, MDA-5, etc).
- Patients should be stratified for cancer as high, intermediate or low risk (see table below)
- All should receive country, age and sex appropriate screening measures - basic screening (eg, history and exam, labs, ESR, CRP, UA, CXR, SPEP). Patients in intermediate and high risk groups should undergo enhanced screening and those with high risk should undergo enhanced screening every 3 years. Enhanced screening measures include gender specific assessments (CT scans, cervical screening, mammography, vaginal US, CA125, fecal occult blood) (all of these are "strong" recommendations).
- Conditional recommendations include:
- FDG/PET-CT for high risk patients in whom nothing is found or patients who are positive for TIF-1gamma, over age 40 years and have >1 high risk feature.
- Endoscopy: upper and lower GI endoscopy for high risk patients in whom nothing is found, endemic areas of nasopharyngeal cancer and in IIM patient with high risk symptoms (weight loss, smoking, fever, night sweats, +Family history)
Editor's note: while I applaud this effort to aid in objective, assessments with reasonable yield, I also believe that this needs to be studied for its true effects and benefits by looking at diagnostic yield and cost efficacy when applied to a suitably large IIM population with age appropriate controls.










If you are a health practitioner, you may Login/Register to comment.
Due to the nature of these comment forums, only health practitioners are allowed to comment at this time.